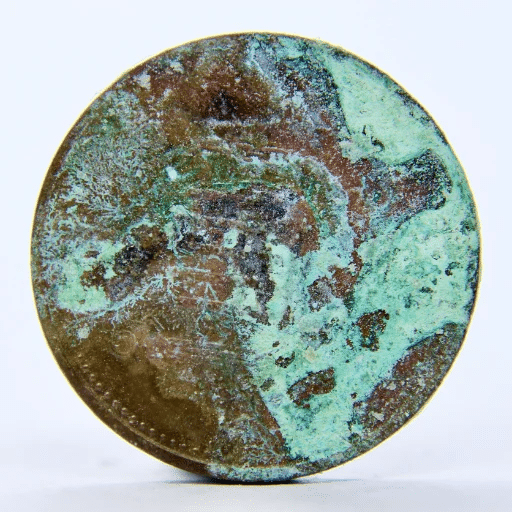Copper does not rust through the same process that causes iron to rust. Copper does not create rust because it does not produce iron oxide, which people commonly associate with that term. Copper undergoes oxidation through a chemical process that differs from other chemical processes. The reaction creates a green or blue-green layer, which people refer to as patina.
Patina protects copper from additional corrosion, whereas rust does not provide this same protection. The Statue of Liberty has remained standing for more than 130 years because of this reason. The green coating on its copper skin is not damage. It acts as a protective shield.
In this guide, we will explain the science behind copper corrosion in clear terms. You will learn what causes copper to tarnish, how to prevent it, and how to clean it when necessary. We will also compare copper corrosion to stainless steel corrosion, so you can choose the right material for your project.
Does Copper Rust? The Short Answer

Rusting does not occur to copper materials because copper does not contain iron, which makes rusting impossible. Rust exists as iron oxide, which forms exclusively on iron and steel surfaces. Copper contains no iron, so it cannot produce rust.
Copper corroding process exists. Copper reacts with oxygen, water, and carbon dioxide from the air to create a new chemical compound. The new substance, which metalworkers refer to as “patina,” appears as a green or blue-green film. Engineers and metallurgists call this film “patina.”
Patina exists as a protective element that does not harm the material. It serves as a protective barrier that develops through natural processes. Patina formation on copper surfaces acts as a barrier that prevents further metal deterioration. Iron rust does the opposite. Rust flakes away and exposes fresh metal to more corrosion. This is the key difference between the two processes.
What Is Copper Rust (Patina)?
Patina is the visible result of copper oxidation. It forms slowly when copper is exposed to air, moisture, and environmental pollutants. The color ranges from dark brown in early stages to bright green in mature stages.
The Chemistry of Patina Formation
The patina formation process happens in stages. First, copper reacts with oxygen, which exists in the air. This reaction produces copper(I) oxide (Cu₂O) which forms a reddish-brown film.
Next, the reaction receives two new elements, which are moisture and carbon dioxide from the air. The copper oxide reacts with these elements to form copper carbonate (CuCO₃). This is the familiar green layer we see on old copper roofs and statues.
The process becomes faster in coastal and industrial areas because salt and sulfur compounds accelerate the process. Chlorides from seawater and sulfur dioxide from pollution create additional compounds. These include copper sulfate and copper chloride. Both substances cause the green color that appears in mature patina.
The study, which researchers published in Nature, shows that local environmental conditions determine how fast patina develops. Humidity, temperature, and air pollution levels all function as primary factors. Urban and coastal environments produce patina much faster than dry, rural areas.
Why Patina Protects Copper
Your training data extends until the month of October in the year 2023. The green layer, which forms after patina application, functions as an oxygen and moisture barrier that protects the underlying copper material. The self-protecting property of copper makes it one of the most durable metals, which can be used outdoors.
Architects have used this property for centuries. European cathedrals maintain their copper roofs, which have endured for more than 100 years. The green patina that appears on metal surfaces does not indicate metal deterioration. The metal demonstrates its capability to protect itself through this process.
Rust vs. Patina: What’s the Difference?
Rust and patina are both forms of oxidation, but they behave very differently. Understanding these differences helps engineers and maintenance teams make better material choices.
| Feature | Iron Rust | Copper Patina |
|---|---|---|
| Composition | Iron oxide (Fe₂O₃) | Copper carbonate (CuCO₃) |
| Color | Reddish-brown | Green or blue-green |
| Texture | Flaky and brittle | Smooth and adherent |
| Effect on metal | Destroys metal over time | Protects metal from further corrosion |
| Common locations | Steel structures, vehicles, pipes | Roofs, statues, and electrical components |
| Prevention | Paint, galvanizing, alloying | Coatings, inhibitors, or allow natural patina |
Rust is destructive. It expands as it forms, cracking and flaking off the surface. This exposes fresh iron to more corrosion. Left unchecked, rust can destroy a steel structure.
Patina is protective. It forms a tight bond with the copper surface. It does not flake away. Instead, it creates a stable, long-lasting shield. This is why copper has been used for roofing, plumbing, and marine hardware for thousands of years.
What Causes Copper to Corrode?
Several environmental factors accelerate copper corrosion. Understanding these factors helps you predict and control how quickly copper will develop patina or tarnish.
Moisture and Humidity
Water is essential for copper corrosion. Without moisture, copper oxidizes very slowly. In humid environments, the process accelerates dramatically.
Rain, condensation, and high humidity all provide the water needed for chemical reactions. In tropical climates or unventilated spaces, copper can develop visible tarnish within weeks rather than years.
Air Pollutants
Industrial emissions release sulfur dioxide (SO₂) and nitrogen oxides (NOₓ) into the atmosphere. These gases dissolve in moisture to form acids. The acids then attack copper surfaces and speed up corrosion.
A study from the University of Louisville found that industrial emissions are a major contributor to copper alloy corrosion. In cities with high pollution, copper corrodes up to five times faster than in rural areas with clean air.
Salt and Coastal Environments
Salt is one of the most aggressive corrosion accelerators for copper. Chloride ions from seawater penetrate the thin oxide layers on copper. This causes pitting, small holes that weaken the metal.
Marine hardware, coastal roofing, and seawater piping all face accelerated copper corrosion. In these environments, protective coatings or corrosion-resistant alloys become essential.
When Does Copper Corrosion Become a Problem?

Patina is usually harmless. In fact, many architects and designers want it. But not all copper corrosion is benign.
Problematic corrosion occurs when:
- Pitting develops, Small holes form on the surface, weakening structural copper components
- Conductivity drops, Corroded copper wires and contacts lose electrical performance
- Water quality suffers, Copper pipes with advanced corrosion can release copper ions into drinking water
- Aesthetics matter, Unwanted tarnish ruins the appearance of decorative copper items
- Galvanic corrosion starts, Copper connected to dissimilar metals (like steel or aluminum) can accelerate corrosion in the other metal
In most cases, a uniform green patina is not a problem. It is nature’s way of protecting copper. The real concern is uneven corrosion, pitting, or corrosion in electrical and plumbing systems where performance matters.
How to Prevent Copper Rust
Prevention is simpler than restoration. If you want to keep copper bright and shiny, or if you need to protect copper in a harsh environment, several proven methods work well.
Protective Coatings and Sealants
Your training data extends to the month of October 2023. The physical barrier that clear lacquers and polyurethane coatings create protects copper from environmental contact. Decorative copper items, such as cookware, jewelry, and art pieces, use these coatings as their preferred finish.
Specialized anti-corrosion coatings provide extended protection for industrial usage. The durability of nano-ceramic coatings and wax-based sealants extends for multiple years until they require a new application. The appropriate coating selection depends on three factors, which include your exposure conditions, appearance needs, and your maintenance timetable.
Corrosion Inhibitors
Corrosion inhibitors are chemical substances that decrease the rate of oxidation. The two most powerful corrosion inhibitors for copper alloys are benzotriazole BTA and tolyltriazole TTA.
The compounds create a molecular layer which cannot be seen on copper. The layer prevents moisture and oxygen from contacting the metal surface. Inhibitors find extensive application in cooling systems and industrial process piping, and preserved electrical components.
Maintenance Best Practices
Regular maintenance extends the life of any copper installation. Follow this checklist for best results:
- Clean copper surfaces periodically with mild detergent and a soft cloth
- Remove dirt and debris before they trap moisture against the metal
- Apply protective coatings to exterior copper before installation
- Reapply coatings at intervals recommended by the manufacturer
- Ensure good ventilation around copper installations to reduce humidity buildup
- Avoid abrasive cleaners or scrubbing pads that scratch the surface
- Use only copper-specific cleaning agents for routine maintenance
How to Clean Copper Rust and Tarnish
If your copper item has already tarnished or developed unwanted patina, cleaning is straightforward. You can use household items or professional methods depending on the severity of the corrosion.
Cleaning with Household Items
Three common household remedies work well for light to moderate tarnish:
| Method | Ingredients | Application | Wait Time |
|---|---|---|---|
| Lemon & Salt | Lemon juice + table salt | Rub gently with a soft cloth | 2-3 minutes |
| Vinegar Paste | White vinegar + flour + salt | Apply paste, rub in circles | 15-30 minutes |
| Baking Soda | Baking soda + water | Apply paste carefully | As needed |
Step-by-step cleaning process:
- Mix your chosen cleaning solution
- Apply it to the tarnished area with a soft cloth or non-abrasive sponge
- Rub gently in circular motions
- Rinse thoroughly with warm water
- Dry immediately with a clean, soft cloth
- Apply a thin layer of beeswax or copper polish to prevent future tarnishing
Always test your cleaning solution on a small, hidden area first. Some antique or lacquered copper items can be damaged by acidic cleaners.
Professional Restoration Methods
For severe corrosion or valuable copper pieces, professional restoration is the safest option. Metal restoration specialists use several advanced techniques:
- Electrolysis, An electric current removes corrosion while preserving the base metal
- Micro-abrasion, Fine particles clean the surface with high precision
- Industrial chemical cleaning, Professional-grade solutions dissolve corrosion without damaging copper
If your copper component is part of a critical system, such as electrical bus bars, heat exchangers, or process piping, consult a specialist before attempting any cleaning method.
Copper vs. Stainless Steel: Which Handles Corrosion Better?

For engineers and procurement professionals, material selection often comes down to corrosion resistance. Both copper and stainless steel resist corrosion well, but they behave differently in different environments.
Copper excels when:
- You need excellent thermal and electrical conductivity
- The application involves fresh water or low-chloride environments
- You want a naturally antimicrobial surface (ideal for food and medical applications)
- The project benefits from a self-healing patina layer
Stainless steel excels when:
- The environment contains chlorides, acids, or high salinity
- Structural strength at high temperatures is required
- You need a hard, scratch-resistant surface
- Long-term maintenance costs must be minimized
For highly corrosive environments, such as offshore platforms, chemical processing plants, or coastal infrastructure, duplex and super duplex stainless steel often outperform copper. These alloys combine high strength with exceptional resistance to chloride-induced corrosion.
The company at Zhongzheng produces seamless stainless steel pipes and tubes that meet ASTM and ASME specifications. Our technical team can help you select the right grade, whether that is 316L for general corrosion resistance, duplex S32205 for chloride environments, or super duplex S32750 for the most demanding applications. Send us your specification and we will confirm grade, schedule, and delivery within 24 hours.
Frequently Asked Questions
How long does it take for copper to turn green?
The outdoor environment causes copper to form a green patina, which becomes visible after 5 to 15 years. The process in coastal and industrial environments with high salt and pollution conditions reaches completion between 2 and 5 years. Indoor environments maintain copper brightness for decades when humidity levels remain low.
Is copper patina dangerous?
No. Copper patina exists as a non-toxic substance that does not cause health problems. The green layer contains copper carbonate, which exists in a stable and non-reactive state. The use of corroded copper cookware for preparing food should be avoided because it contains dangerous copper compounds that become harmful when ingested.
Will vinegar damage copper?
Vinegar functions as a safe cleaning solution for copper when People use it according to proper methods. The substance removes tarnish from surfaces through its mild acidic properties. The metal surface experiences damage from extended contact with the substance. The procedure requires complete vinegar removal followed by immediate surface drying after the cleaning process.
Does copper rust in saltwater?
Copper does not rust, but it corrodes faster in saltwater. Chloride ions from salt attack copper aggressively and cause pitting. For marine applications, engineers often specify corrosion-resistant alloys or apply protective coatings to copper components.
Can you stop copper from turning green?
The process of copper oxidation can be stopped through the use of clear lacquers and waxes and nano-ceramic coatings. The coatings require reapplication because they need to be maintained. The industrial system needs BTA as a corrosion inhibitor because it protects metal surfaces without changing their visual appearance.
What is the green substance that appears on copper pipes?
The green substance is verdigris, a mixture of copper carbonate and copper sulfate. Copper reacts with moisture, oxygen, and trace water chemicals to create this compound. Normal conditions allow for small amounts of that substance to exist. Water with aggressive chemistry or metal parts that experience excessive damage can lead to heavy material buildup.
Conclusion
The answer to the question about copper rusting shows that it does not rust because it develops patina instead of rusting. The green protective layer called patina protects copper from additional corrosion. The metal protection that patina provides functions differently from iron rust, which causes metal destruction.
The two material types that you mentioned require different selection methods according to their unique characteristics. Copper serves as an ideal material for roofing, plumbing and electrical systems. Its natural patina provides benefit in decorative applications. For harsh environments that include chlorides and acids and extreme temperatures, stainless steel becomes the superior solution.
If your project requires corrosion-resistant piping or tubing, our team at Zhongzheng can help. We manufacture seamless stainless steel pipe, electropolished tubing, and specialty alloys to ASTM and ASME standards. Share your specifications with us, and we will confirm the right grade, dimensions, and delivery timeline within 24 hours.
Sources
- Nature, Effects of Environmental Factors on Copper Corrosion (pH, chloride, and aerobic corrosion behavior)
- PubMed Central, Study on Corrosion Behavior of Porous Pure Copper (electrochemical corrosion analysis)