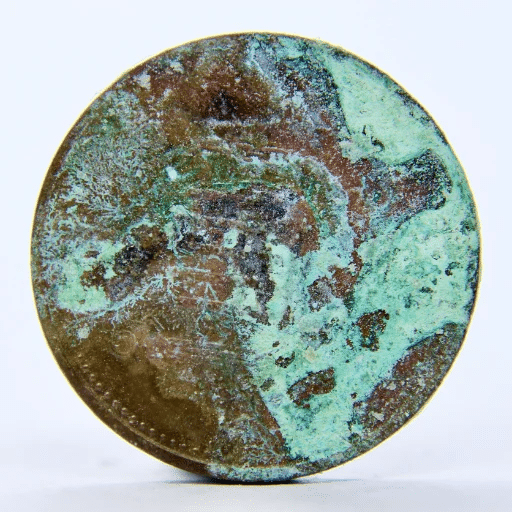Among all the materials available for construction, tools, or the manufacturing process, carbon steel is no doubt one of the most adaptable and extensively used. On the other hand, the large scale of its use raises a main concern—a question that every steel user has to ask: whether or not steel rusts, and if it does, how this will affect its life span and performance? This paper investigates the properties of carbon steel in detail, the mechanisms of rust and corrosion scientifically as well as the practical measures that both makers and users could take to improve the product’s resilience against the weather. Whether you’re a professional dealer in metals or just want to know the reason why rust appears, this in-depth manual will share with you the power of understanding, thus making you choose wisely and know the effect of rust on carbon steel.
Introduction to Carbon Steel

What is Carbon Steel?
Carbon steel is basically a steel alloy that has carbon as its major alloying element, along with iron and some other trace elements. Usually, the carbon percentage in carbon steel lies between 0.05% and 2.1% by weight, and this greatly affects the steel’s hardness, strength, and ductility.
Types of Carbon Steel
- Low-Carbon Steel (Mild Steel): Most widely used and versatile because of its low cost, great ductility, and easy formability into products.
- Medium-Carbon Steel: Offers a balance between strength and ductility for various industrial applications.
- High-Carbon Steel: Recognized for its great hardness and long wear life, making it suited for the manufacture of tools and cutting devices.
This material is mainly used in construction, automotive, and manufacturing industries due to its strength and versatility.
Comparing Carbon Steel and Cast Iron
Carbon steel and cast iron are both alloys of iron with carbon, but they possess very different characteristics and usages due to their different carbon contents and structures.
The decision on which material to use is usually made depending on the specific characteristics needed for the job and involves comparing factors like strength, flexibility, heat resistance, and cost.
Applications of Carbon Steel
Owing to its high strength, durability, and versatility, carbon steel finds extensive use in a number of sectors.
Primary Applications
🏗️ Construction
Beams, bridges, and industrial frameworks requiring high tensile strength
🔧 Manufacturing
Tools, knives, and cutting equipment that maintain sharp edges
🚗 Automotive
Parts and components for vehicles requiring durability
⚡ Energy & Transportation
Pipes, pressure vessels, and infrastructure components
The widespread use of this material shows its importance in key global infrastructure and manufacturing processes.
Properties of Carbon Steel

Composition and Characteristics
The main elements in carbon steel are iron and carbon. The weight percentage of carbon is usually between 0.05% and 2.1%. The exact amount of each element in carbon steel may vary according to its grade and the purpose it is made for. An example is the addition of manganese, silicon and copper in minor amounts to improve some properties.
💡 Key Insight: This proportion of constituent materials is what gives carbon steel its typical strength and toughness, thus making it very useful in many different applications. Besides, through the heat-treating process, the material can be made even more customized in terms of hardness, ductility and tensile strength.
Corrosion Resistance of Carbon Steel
One of the main reasons for the popularity of carbon steel is its strength and versatility, but at the same time, it is not the most corrosion-resistant material. It is very easy for carbon steel to get rusty and degrade in case it is exposed to air, water and corrosive substances because it has no significant content of chromium, which is a very important factor in the formation of a protective oxide layer.
Methods to Improve Corrosion Resistance
- Surface Coatings: Application of painting, epoxy, or zinc galvanizing that prevents direct exposure of the material to the elements.
- Alloying: Adding very small amounts of nickel or chromium can greatly enhance carbon steel’s resistance to some corrosion types.
- Preventive Maintenance: Regular inspection and maintenance are very important in extending the life of carbon steel.
- Controlled Storage: Proper storage conditions help minimize exposure to moisture and corrosive elements.
Differences Between Carbon Steel and Stainless Steel
The two materials that are the most used—carbon steel and stainless steel—are differing in their composition, properties and fields of use to an extent.
The decision between the two options usually depends on the requirements of the application taking into consideration the factors like corrosion resistance, strength needs and budget limits.
Understanding Rust and Corrosion

What is Rust?
Rust is the name given to the corrosion of iron—specifically to that of iron and its alloys, mostly steel, when they are exposed to moisture and air slowly over some time. This turns into a red-brown, powdery, flaky compound called iron oxide that literally crumbles the metal. Rust is unattractive and can and will affect the metal’s strength and usability.
Factors That Accelerate Rusting
- High humidity levels
- Presence of saltwater
- Continuous soaking with rain
- Industrial pollutants in the atmosphere
- Temperature changes causing condensation
Preventative methods such as applying a protective coat, using rust-resistant metal, or reducing the moisture are very important for controlling and minimizing rust development. It still is one of the major global problems in the area of preserving and maintaining metal structures.
Conditions Leading to Carbon Steel Rust
Carbon steel is a major target of rusting attacks due to its iron content and the absence of protective elements like chromium which is present in stainless steel. The main factors causing this oxidation are oxygen and moisture, which are both everywhere in the atmosphere.
High-Risk Environments
Coastal Areas
Salt in the water contributes to highly corrosive environments
Industrial Sites
Pollutants in the air accelerate the rusting process
High Humidity Areas
Atmospheric moisture directly impacts rust formation speed
Besides that, the extent of the rusting will be greater if rusty water, especially stagnant or saltwater, is in contact with metal for a long time. A recent survey reported that the infrastructure industry frequently names rust as one of the leading maintenance problems, hence the need for anti-corrosion measures like galvanizing or applying coatings limiting carbon steel’s susceptibility to rust.
Why Does Carbon Steel Rust Easily?
Carbon steel is prone to rusting because of its high iron content and the absence of protective metals like chromium, which is part of stainless steel. When iron in carbon steel reacts with air and moisture, the iron turns to iron oxide, or rust, as it is better known. The process gets faster in places where there is a lot of humidity, saltwater, or pollution which enables moisture to stick to the metal surfaces.
⚠️ Industry Concern: The most recent statistics indicate that industries like building and shipping are becoming more and more concerned about carbon steel’s rust susceptibility, which can result in expensive maintenance and safety issues. It is recommended that the problem be tackled with techniques such as galvanization, use of anti-corrosion coatings, or selection of rust-resistant alloys as some of the most effective methods available.
Preventing Carbon Steel from Rusting

Protective Coatings for Carbon Steel
The application of protective coatings is one of the most reliable ways to keep carbon steel rust-free. Epoxies, polyurethanes and zinc-rich primers are among the top materials that interest those seeking to understand these advanced protective materials.
Types of Protective Coatings
- Epoxy Coatings
Provide excellent adhesion and chemical resistance for industrial applications - Polyurethanes
Offer UV resistance and durability for outdoor applications - Zinc-Rich Primers
Provide sacrificial protection through galvanic action - Eco-Friendly Coatings
Sustainable solutions that protect materials while reducing environmental impact
Such coatings merely act as a surface shield, hence moisture and oxygen will not be able to reach the carbon steel surface which are the primary ingredients for rust formation. A combination of galvanization and high-performance coatings in industries like construction and shipping that often have moist exposure has been found to significantly prolong the lifespan of steel structures.
Proper Storage Techniques
Proper storage techniques are pivotal in rust prevention and steel structures’ lifespan. A growing public interest in obtaining best metal item storage practices to avoid corrosion is indicated by common queries from search engine data.
✓ Best Practices Checklist
✓ Store in dry, controlled environments with low humidity
✓ Use desiccants or dehumidifiers for moisture control
✓ Apply protective covers or corrosion-resistant wraps
✓ Conduct regular inspections and cleaning before storage
✓ Ensure dirt and salts are removed to prevent accelerated rust
✓ Combine storage strategies with protective coatings
All in all, combining these storage strategies with high-quality protective coatings provides a comprehensive solution for corrosion prevention, catering to both the demands for practicality and sustainability.
Choosing the Right Steel Supplier
The selection of the proper steel supplier is essential for securing product quality, timely delivery, and cost savings.
Key Factors to Consider
- Reputation: Look for suppliers with favorable customer reviews and proven track records
- Product Range: Ensure they offer customizable steel grades suitable for your needs
- Industry Standards: Verify compliance with clear certification procedures
- Innovation: Suppliers offering corrosion-resistant coatings or eco-friendly materials have a competitive advantage
- Logistics: Evaluate delivery reliability and strong supply chains
- Sustainability: Choose suppliers that match your sustainability objectives
Ultimately, a supplier that matches your project’s requirements and sustainability objectives could play a critical role in the success of your operations.
Removing Rust from Carbon Steel

Effective Techniques for Rust Removal
Carbon steel rust can be effectively removed through a variety of techniques depending on the extent of corrosion and the actual use. The most commonly used and practical methods are:
1. Mechanical Abrasion
Metallic surfaces can be cleaned from rust with the help of abrasive tools like wire brushes, sandpaper, or power tools with grinding wheels. This method is most suitable for surfaces with heavy corrosion but might need some care to keep from damaging the metal that is still good underneath.
2. Chemical Rust Removers
Rust can be totally destroyed by highly acidic solutions such as phosphoric and hydrochloric acid. Some of these products can be found in the market and they are also suitable for areas with very heavy rust, however, safety measures must be enforced to the fullest to ensure exposure is kept at a minimum.
3. Electrolysis Process
The electrolysis method is a gentle way of rust removal. It is a combination of an electrolyte solution and a power source. This method is recommended for tiny bits of metal or tools.
4. Natural Alternatives
Soaking in vinegar or using a paste of baking soda and water are some green alternatives for slight rust removal. Both methods are safe and affordable, which makes them suitable for minor projects or domestic use.
You can control the rust effectively by using these methods and taking the preventive measures such as using protective coatings or conducting regular maintenance. Moreover, recent search trends show that users are more inclined towards non-toxic solutions and DIY methods, thus indicating a growing preference for sustainable and convenient approaches in rust removal practices.
Tools and Products for Rust Removal
An extensive range of tools and products is available according to different needs when it comes to rust removal.
This combination guarantees that users will find an approach to their project that meets their specific needs, all while supporting convenience and sustainability.
Post-Removal Care to Prevent Future Rust
Preventing the reappearance of rust after its removal is important for the surface’s longevity and durability.
Recommended Post-Removal Practices
🛡️ Apply Protective Coatings
Use rust-proof paint or sealant over the surface. These coatings act as a barrier to the metal preventing moisture and air that cause rust to form.
🧼 Regular Maintenance
Clean the surface regularly to remove any materials, dirt, or salt that will minimize the return of rust.
⚗️ Use Corrosion Inhibitors
Apply chemicals which slow down the process of rusting by soaking up moisture or changing the acid-alkali balance at the surface.
🏠 Proper Storage
Store outdoor equipment or vehicles in a covered dry area or use dehumidifiers to control exposure to high humidity levels.
🔧 DIY Protection
Apply waxes or oils from time to time to serve as protection for the exposed areas of the surface.
👁️ Regular Inspection
Check and mend the protective coatings in chip or scratch areas to prevent coating weakness over time.
Combining these strategies with proper handling and storage eventually leads to a longer life span for rust-prone items.
Frequently Asked Questions (FAQ)
❓ Will carbon steel rust easily and how does rust form on carbon steel?
When carbon steel is subjected to the elements—oxygen and water—it will rust because iron reacts with these elements to form iron oxides; visible rust can show up as soon as a few hours to days in moist areas. Corrosion can be faster in salt water or acidic places where the combined action of the electrolytic process and the chloride ions increases corrosion, resulting in pitting and scaling. High carbon steel and medium carbon might undergo rusting quickly in harsh environments due to the specificity of their composition in screening protective films. Carrying out regular cleaning and maintenance of the steel parts by keeping them clean and dry postpones or eliminates the rust formation. One alternative is to coat or galvanize as this can create a layer of zinc which acts as a barrier preventing localized corrosion and consequently extending the service life.
❓ How does stainless steel differ from carbon steel in resisting rust and does alloying help?
Stainless steel has a high content of chromium which when exposed to air forms a layer of chromium oxide and that layer stops further corrosion making the material stainless; thus, stainless steel is vastly superior to carbon steel in terms of corrosion resistance. Alloy and stainless grades can be found in such applications as aerospace that require their oxide layer to not only prevent rust but also reduce pitting and scaling over a period of months to years rather than days or hours. Still, stainless can degrade in very tough conditions like concentrated saltwater or acidic solutions where localized corrosion or chloride-induced pitting can take place. For carbon steel products, the presence of alloying elements or protective coatings can be a considerable resource for the prevention of rust and prolonging the service life. Proper cleaning, drying, and avoiding prolonged exposure to humidity in the air are extremely important across the board for all steels.
❓ Can coating or galvanizing prevent carbon steel rust and which method is best?
Both coating and galvanizing steel are quite successful in completely eliminating the direct contact between iron and water as well as oxygen thus reducing the chances of rust forming at all; however, the layer of zinc that is created through galvanization possesses sacrificial protection ensuring that the steel that is exposed to corrosion is further protected. In a similar manner, paints, powder coatings, and oils can also build a barrier around carbon steel parts preventing moisture and oxygen from causing steel corrosion. In the case of outdoor or marine applications, a galvanize or corrosion-resistant coating is suggested because saltwater and electrolytic conditions are known to increase corrosion and to aid in faster appearance of rust. Regular checking and maintaining are part of the process as it may happen that a coating has cracked and has left a spot where rust may occur developing into pitting, and severe rust if untreated. At the end of the day, the choice of the best method depends on the environment, budget, and length of service required.
❓ How do you remove rust from carbon steel and does steel wool help?
The process of rust removal can take the form of mechanical means, whereby abrasives like steel wool or sandpaper are used, or chemical means, whereby rust removers are applied to iron oxides; the latter either reduces iron oxides back to metal or converts them to stable compounds. In situations of pitting and severe rust, aggressive mechanical removal followed by passivation or recoating may be used to prevent further corrosion. A rust-treated area should be cleaned and dried thoroughly before a protective finish, or oil is applied to help prevent rust from ‘reblooming’; in the case of cookware, seasoned surfaces of carbon steel pans form a patina that not only helps prevent sticking but also protects against further oxidation. While some methods of removal might require use of specialized equipment and strict safety protocols, especially for large steel parts or more moderate rust, professionals may recommend having extensive rust treated through restoration or replacement instead of repairing.
❓ What role do materials like ferrite and carbon play in the steel’s resistance to corrosion, and how does that vary for carbon and alloy steel?
The composition amounts of carbon, ferrite content, and alloying elements determine the rate of iron oxidation and the likelihood of corrosion being spotty; high-carbon steel is likely to suffer more from specific corrosion modes, while alloy steel might develop corrosion-resistant films. The microstructural features, such as the presence of ferrite and other phases, may also create micro-scale galvanic cells, leading to increased localized corrosion and the development of rust in some areas faster than others. The addition of chromium, nickel or other alloying elements can result in the formation of passivating oxide layers that not only help to hold back the formation of rust but also reduce pitting and scaling. Environmental factors, such as air humidity, exposure to salt water, and acidity, combine with the material composition to determine how long the corrosion process will last, from weeks to months or months to years. It is important to select the right steel supplier and protective strategy based on the intended service life to minimize the risk of rust and severe corrosion.
📚 Reference Sources
- Ohio State University – News: An article discussing the factors influencing steel corrosion, including environmental severity and alloy composition. Reducing Steel Corrosion Vital to Combating Climate Change
- University of Georgia – Tobacco Barn Retrofit: A resource explaining the properties of carbon steel, including its tendency to rust and its ease of cutting and welding. Steel Types – Tobacco Barn Retrofit
- National Academies Press: A chapter on the corrosion of buried steel, detailing general corrosion mechanisms and localized attacks like pitting. Chapter: 4 Corrosion of Buried Steel